Hershey Lab
Projects
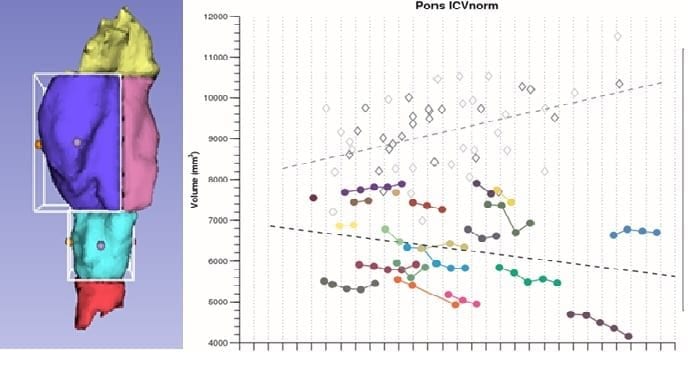
The Wolfram Clinic
The goal of the Wolfram Clinic is to better understand the development and progression of the neurological aspects of Wolfram syndrome. During annual research visits patients are seen by specialists in multiple disciplines (ophthalmology, neurology, psychiatry, audiology) and undergo magnetic resonance imaging (MRI) of the brain. This data helps us understand the neurological issues in Wolfram syndrome, and will directly inform the design and metrics used in future clinical trials.
Diabetes: The MetaBrain Study
Type 2 diabetes mellitus (T2D) is a significant public health problem affecting ~30 million Americans. Obesity, insulin resistance, insulin deficiency (β cell dysfunction) and dysglycemia all precede the diagnosis of T2D and are known to promote inflammation and ultimately lead to microvascular complications. More recently, research has identified brain-related complications in adult-onset T2D, including reduced regional brain structure and function, impaired cognition, and increased lifetime risk for Alzheimer’s disease. Alarmingly, an increasing number of children and adolescents are being diagnosed with T2D, likely due to the growing prevalence and earlier onset of obesity. Youth-onset T2D appears to have a more aggressive course than adult-onset T2D, with earlier onset and more rapid progression of microvascular complications. In addition, studies of youth with obesity and youth-onset T2D have reported robust differences in regional brain structure and cognition, suggesting that brain effects may follow the same aggressive course as the more typical vascular complications. Unfortunately, little is known about the factors associated with poor brain structure and function in youth with T2D.
Diabetes: The NewT Study
The Hershey Lab and collaborators are studying how the brain is a target organ in diabetes. In particular, we are interested in how brain development and exposure to hyperglycemia and hypoglycemia interact to shape developmental trajectories of the brain and its functions. This work has led to an understanding of the regional brain vulnerability in diabetes and has been funded for the past 10 years by the NIH (NIDDK).
Enrollment for study has ended; analyses are ongoing.
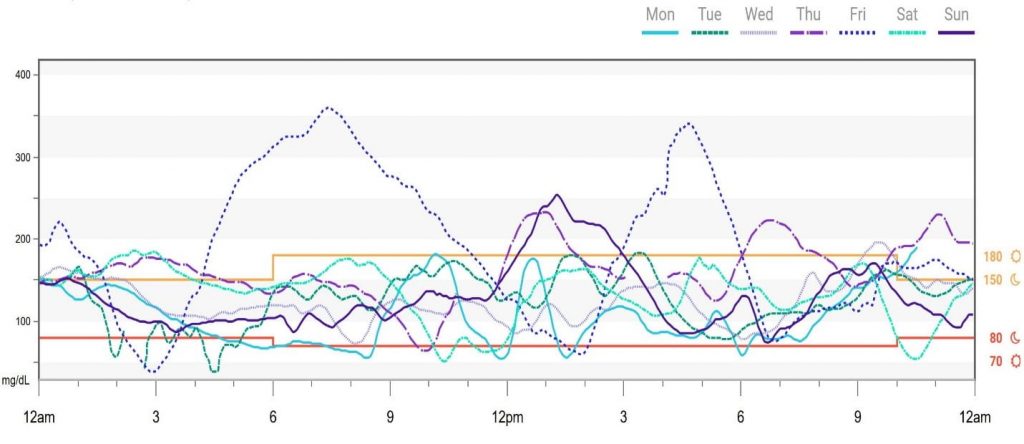
Diabetes: Pediatric Ambulatory Research in Cognition (PARC) Study
Management of blood glucose with type 1 diabetes (T1D) is challenging for youth and often results in frequent swings between normal, high, and low glucose (Awoniyi, 2013) like that shown in Figure 1. This glucose variability remains poorly understood, but research has shown that it is a potential risk factor for diabetic complications (Ceriello, 2019). One of the least understood complications of T1D is cognitive impairment. Several studies have shown impaired cognitive function in youth with T1D compared to their non-diabetic peers, and that poorer glycemic control including severe glycemic events (e.g., severe hypoglycemia) and chronic hyperglycemia are associated with cognitive impairments (Cato 2016). These studies assessed the important relationship between long-term glucose variability in T1D and cognition; however, little is still known about how cognitive function is impacted daily in real-life settings, particularly with dynamic cognitive skills that fluctuate throughout the day (e.g., working memory) (Gamaldo, 2016).
Obesity: Neuroimaging-Based Measures of Putative Brain Inflammation
Obesity is a major risk factor for diabetes and late-life cognitive impairment and dementia, including Alzheimer disease. As longevity rises and the rate of obesity increases globally in children and adults, it is important to understand mechanisms by which obesity confers risk for late-life disease. One such mechanism may be obesity-related brain inflammation, or neuroinflammation.
Obesity
Human obesity is a major public health problem that is driven by a complex combination of behavioral, genetic, environmental, biological and neurobiological factors. Neuroimaging studies in humans find altered dopamine (DA) function and reward-related behavior associated with obesity but conflicting findings limit our understanding of these complex relationships.
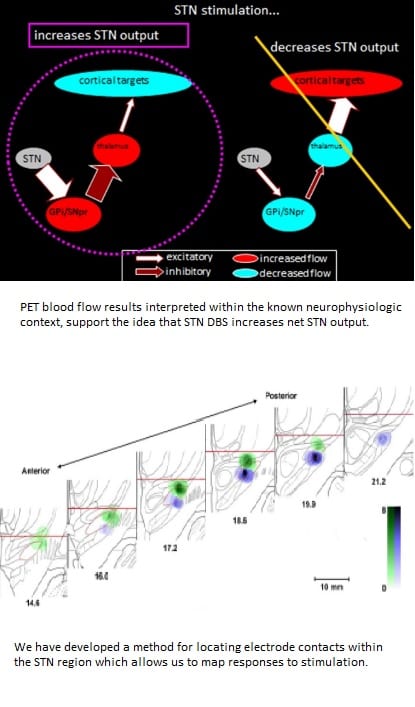
Parkinson’s Disease
Deep-brain electrical stimulation (DBS) can provide substantial motor benefit and impact non-motor domains of cognition and mood in Parkinson’s disease (PD) when implanted in the subthalamic nucleus (STN). However, the neural mechanisms underlying these effects are not fully understood. We are collaborating with the Culver Lab to use high density diffuse optical tomography (HD-DOT) to understand how STN DBS alters functional cortical networks in the brain.